Steroid Sans Chemotherapy in a Case of Tuberculous Pleural Effusion: A Novel Proposition or a Recipe for Disaster?
Gyanshankar P. Mishra1*, Ninu P. Babu2
1Dept. of Respiratory Medicine, Indira Gandhi Government Medical College, Nagpur, Maharashtra, India
2Dept. of Respiratory Medicine, Indira Gandhi Government Medical College, Nagpur, Maharashtra, India
Abstract
Tubercular pleural effusion is one of the extrapulmonary forms of tuberculosis. We herein report the case of a thirty-five-year-old female diagnosed with Tubercular pleural effusion who inadvertently took steroid monotherapy for four weeks sans antitubercular chemotherapy. On follow up she showed clinico radiological improvement. The case report discusses the potential role of steroids in current management of tubercular pleural effusion.
Introduction
TB (Tuberculosis) is a communicable disease that is the leading cause of death from a single infectious agent. Typically, the lungs are affected (pulmonary TB) but other sites may also be affected (extrapulmonary TB) in TB. About a quarter of the world’s population is infected with Mycobacterium tuberculosis and thus at risk of developing TB disease1.
Case Report
A thirty-five years old female patient presented with complaints of dry cough, fever, right sided chest pain and exertional breathlessness since last 10 days. She did not have any associated co-morbidities and denied any past history of TB or contact with a TB case. On examination, patient weighed 50 kgs and physical examination revealed decreased breath sounds on the right side.
TLC (Total Leukocyte Count) was 10,600/mm3, DLC (Differential Leukocyte Count) was: Polymorphs: 68 %, Lymphocytes: 23 %, Eosinophils: 2 %, Monocytes: 3 % and Hb (Hemoglobin) was 11.9 g/dl. The liver function tests and renal function tests were within normal limits. She was seronegative for HIV (human immunodeficiency virus) and HBsAg (hepatitis B surface antigen). Chest Radiograph showed blunting of right costophrenic angle, suggestive of right pleural effusion (Figure 1). USG (ultrasonography) of the thorax revealed mild collection in right pleural space and no collection in left pleural space. USG abdomen was within normal limits. Diagnostic thoracocentesis revealed the following pleural fluid examination findings: ADA (adenosine de aminase) - 54.2 U/L (increased), Protein- 4.30 gm/dl, Glucose- 58 mg/dl, TLC – 1200 cells/cumm, Differential cell count: 80% lymphocytes and 20% polymorphs, LNR (lymphocyte-neutrophil ratio): 4, Gram stain- no organism seen, ZN (Ziehl-Neelsen) stain for AFB (acid fast bacilli) was negative, CBNAAT(cartridge based nucleic acid amplification test) was negative for tuberculosis, cytology and cellblock examination did not reveal any evidence of malignancy. Her induced sputum was also negative for AFB smear and Gram stain and culture.
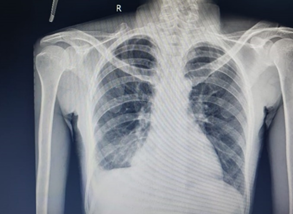
Figure 1: Chest Radiograph at diagnosis showing blunting of right costophrenic angle suggestive of Right Pleural effusion.
Patient was diagnosed as a case of right tubercular pleural effusion, categorized as clinically diagnosed extrapulmonary TB under the NTEP ( National TB Elimination Program) in India and was advised NTEP programmatic management of TB with daily ATT (Anti Tubercular Treatment) regime comprising of four drugs (Isoniazid, Rifampicin, Ethambutol and Pyrazinamide) for two months followed by three drugs (Isoniazid, Rifampicin and Ethambutol) for four months. Patient was also advised adjuvant corticosteroid in tapering doses to hasten symptomatic and radiological improvement2. Patient received the first anti TB drug dose in ward. Patient was discharged on ATT accompanied with tapering doses of oral corticosteroids for four weeks (Tablet prednisolone 20 mg OD (once a day) for seven days, followed by tablet prednisolone 15 mg OD for seven days followed by tablet prednisolone 10 mg OD for seven days, finally tapered to tablet prednisolone 5 mg OD for seven days and then stopped).
Patient came for follow up after five months. She was asymptomatic and on detailed history it was surprisingly found that she was taking steroids as advised, but without ATT. Fresh chest radiograph was done (Figure 2) and it was suggestive of radiological resolution of right pleural effusion. Confirmation of resolution was done by a fresh USG of thorax which revealed no evidence of fluid collection in either pleural cavity.
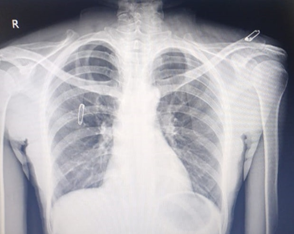
Figure 2: Chest Radiograph at 5 months follow up suggestive of radiological resolution of right pleural effusion.
Patient was offered a fresh course of ATT considering the possibility of relapse in future, but she refused the same and agreed to be in a regular clinical follow up on an OPD (Outdoor patient department) basis.
Discussion
Our patient presented with clinico-radiological features of tubercular pleural effusion confirmed on diagnostic thoracocentesis. In patients with lymphocytic pleural effusion (lymphocyte to neutrophil ratio > 0.75), pleural fluid ADA levels of greater than 40 U/L are suggestive of a diagnosis of TB pleuritis, especially in areas with a high prevalence of tuberculosis3. In fact, in high TB endemic areas or situations of recent exposure of the patient, a lymphocyte predominant exudate with a high ADA with or without granulomas on a pleural biopsy specimen is sufficient evidence to initiate treatment4. Thus, tubercular pleural effusion was the most likely diagnosis in our patient. Without treatment, TB pleuritis is likely to resolve spontaneously; however, the patient frequently develops active TB at a later date5.
Under the programmatic management of TB in India, cases are categorized into two groups viz. microbiologically confirmed TB and clinically diagnosed TB. Microbiologically confirmed TB refers to a presumptive TB patient with biological specimen positive for Acid Fast Bacilli or positive for Mycobacterium tuberculosis on culture or positive for tuberculosis through quality assured rapid diagnostic molecular test. Clinically diagnosed TB case refers to a presumptive TB patient who is not microbiologically confirmed, but has been diagnosed as active TB by a clinician on the basis of X-Ray abnormalities, histopathology or clinical signs with a decision to treat the patient with a full course of ATT. Our patient was therefore classified as clinically diagnosed extrapulmonary TB.
The literature showcasing evidence for use of corticosteroids in TB pleural effusion is conflicting. Corticosteroids induce their anti-inflammatory effect through the regulation of gene expression in cells, leading to increased expression of genes which inhibit inflammatory pathways, and repression of genes encoding pro-inflammatory proteins. The theoretical basis for using corticosteroids is that they suppress the delayed type hypersensitivity inflammatory response triggered by the release of tubercular antigens into the pleural space. While the inflammatory response is necessary to control the infection, excessive inflammation can lead to tissue damage and fibrosis, which could cause long-term morbidity. Suppression of the inflammatory response in pleural TB could reduce the symptoms and signs associated with the inflammatory process: fever, progression of the pleural effusion and malaise. Corticosteroids could therefore reduce the duration or severity of symptoms in the short term and reduce the risk of tissue damage leading to lung impairment in the long term2.
As per latest India TB report, HIV co-infection among TB was nearly fifty thousand cases amounting to TB-HIV coinfection rate of 3.4%.6 However in all combined studies of the Cochrane review published in 2017, on the role of steroids in TB pleural effusion, the proportion of TB-HIV confection rates were a minimum of 33% to a maximum possible 68%, which is approximately at least 10 times the rates seen in Indian population. In spite of this huge difference in the proportion of HIV patients, the Indian index TB guidelines relied on the findings of the Cochrane review to formulate recommendations against overall use of corticosteroids in all patients of TB pleural effusion7. This is especially important considering the fact that HIV-TB patients have a poor TB treatment outcome and are more prone to risk of adverse events to anti-TB drugs as well as corticosteroids2,8. In the study by Wyser et al. (1996), which excluded PLHIV (people living with HIV), no serious ADR (adverse drug reactions) were noted with use of adjunctive steroids in tubercular pleural effusion9. Thus, applying the results of such an analysis in management of TB pleural effusion patients not suffering from HIV requires review. The same Cochrane review of 2017, with six trials and 590 participants, showed that corticosteroids reduced the risk of pleural abnormalities, such as pleural thickening or pleural adhesions in almost one-third of patients. This finding points to an absolute risk reduction of 16%, which is equivalent to a number necessary to treat of seven patients for one individual benefit2,10. Thus, corticosteroids may reduce the time to resolution of the symptoms of TB pleurisy and the time to resolution of the pleural effusion on chest X-ray. Corticosteroids may also reduce the risk of having signs of pleural scarring on chest X-ray (pleural thickening and pleural adhesions) after the disease has resolved2.
There may be a subset of patients diagnosed with tubercular pleural effusion, who may benefit from adjunctive use of corticosteroids. These may be patients who present with severe systemic symptoms (e.g., fever, malaise, pleuritic pain) after therapeutic thoracentesis and after two weeks of treatment, a short period of corticosteroids may be beneficial10. Our patient had right sided chest pain which could have been suggestive of initial pleuritis and thus may have benefited by the anti-inflammatory effects of steroid. In patients with diabetes mellitus, uncontrolled hypertension, peptic ulcer disease, empyema, HIV and neoplastic disease adjunctive corticosteroids may not be used9.
The current case demonstrates the potential role corticosteroids could have in treatment of Tubercular Pleural effusion in non-HIV patients especially as an add on with ATT. In the current case, corticosteroid therapy even in absence of ATT did not lead to flare up of disease and this possibly points to the fact that there may still be some gaps in our understanding of the complex pathophysiological processes involved in genesis of tubercular pleural effusion. Apparently, rather than the bacillary burden, it is the hypersensitivity inflammatory response that is at play towards the pathophysiology of disease process in such patients. This hints towards a potential utility of adjunctive therapeutic targeting of the later process also, whereas the current chemotherapeutic approach specifically targets only the former. Thus, the current recommendations against overall use of adjunctive corticosteroids with ATT in all tuberculosis pleural effusion also needs to be reviewed as there may be a select group of patients in whom it may be of utility. In today’s era of evidence-based medicine, moving towards individualised therapy, the one size fits all approach needs to end and careful evaluation of patients with TB pleural effusion for adjunctive steroid therapy may benefit them.
References
- World Health Organisation. Global Tuberculosis Report 2019 [Internet]. Geneva, Switzerland; 2019. Available from: https://apps.who.int/iris/bitstream/handle/10665/329368/9789241565714-eng.pdf?ua=1
- Ryan H, Yoo J, Darsini P. Corticosteroids for tuberculous pleurisy. Cochrane database Syst Rev [Internet]. 2017 [cited 2019 Oct 6]; 3(3): CD001876. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28290161
- Zhai K, Lu Y, Shi HZ. Tuberculous pleural effusion. J Thorac Dis [Internet]. 2016 Jul [cited 2019 Oct 6]; 8(7): E486-94. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27499981
- Shaw JA, Irusen EM, Diacon AH, et al. Pleural tuberculosis: A concise clinical review. Clin Respir J. 2018 May 1; 12(5): 1779–86.
- Cohen LA, Light RW. Tuberculous Pleural Effusion. Turkish Thorac J [Internet]. 2015 Jan [cited 2019 Oct 5]; 16(1): 1–9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29404070
- Central TB Division (Ministry of Health and Family Welfare). India TB Report 2019: Revised National Tuberculosis Control Programme Annual Report [Internet]. Delhi, India; 2019. Available from: https://tbcindia.gov.in/WriteReadData/India TB Report 2019.pdf
- Sharma SK, Ryan H, Khaparde S, et al. Index-TB guidelines: Guidelines on extrapulmonary tuberculosis for India. Indian J Med Res [Internet]. 2017 Apr [cited 2019 Oct 6]; 145(4): 448–63. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28862176
- Desai C, Shah A, Dikshit R, et al. Efficacy and safety of anti-tuberculosis drugs in HIV-positive patients: A prospective study. Indian J Pharmacol [Internet]. 2013 [cited 2019 Dec 10]; 45(5): 447. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24130377
- Wyser C, Walzl G, Smedema JP, et al. Corticosteroids in the treatment of tuberculous pleurisy: A double- blind, placebo-controlled, randomized study. Chest. 1996; 110(2): 333–8.
- Antonangelo L, Faria CS, Sales RK. Tuberculous pleural effusion: diagnosis & management. Expert Rev Respir Med [Internet]. 2019 Aug 3; 13(8): 747–59. Available from: https://doi.org/10.1080/17476348.2019.1637737
